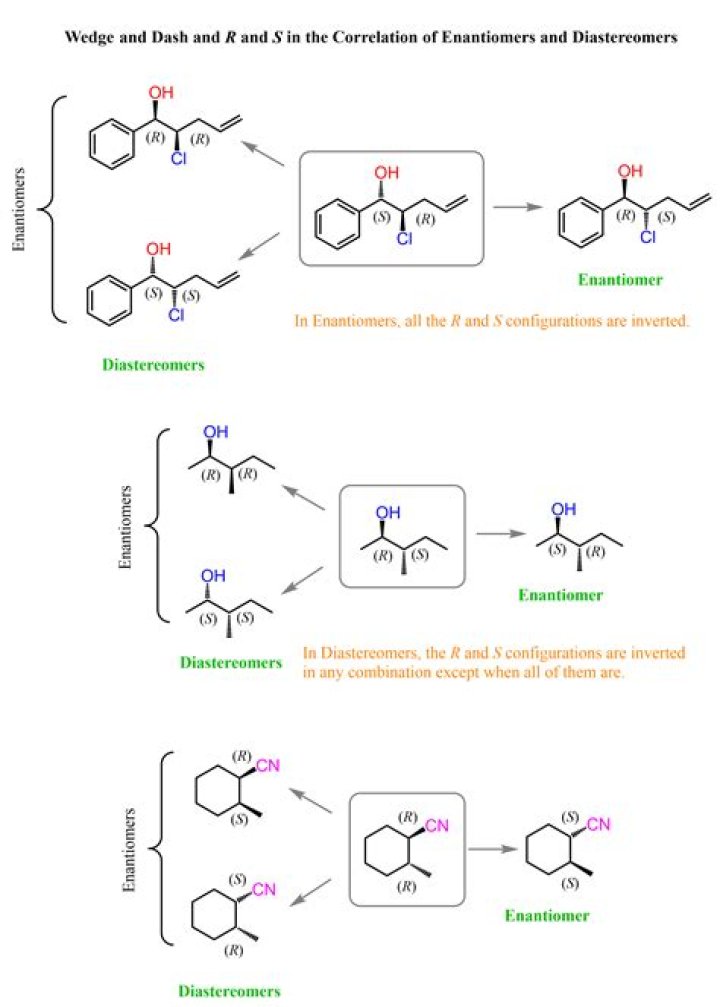
Stereoisomers that differ at some stereocenters but not at others are not mirror images, so they are not enantiomers. Instead, they are diastereomers. A diastereomer is simply any stereoisomer that is not an enantiomer.
Are all diastereomers stereoisomers?
EnantiomersDiastereomersEnantiomers have one or more stereocenters.Diastereomers have two or more than two stereocenters.
Are all stereoisomers enantiomers or diastereomers?
Stereoisomers always have the same connectivity. Among molecules with the same connectivity: Molecules that are mirror images but non-superimposable are enantiomers. If they aren’t superimposable, and they aren’t mirror images, then they’re diastereomers.
Are diastereomers and stereoisomers the same?
Diastereomers are stereoisomers that are not related as object and mirror image and are not enantiomers. Unlike enatiomers which are mirror images of each other and non-sumperimposable, diastereomers are not mirror images of each other and non-superimposable.Are enantiomers the same as stereoisomers?
Enantiomers are stereoisomers that are non-superimposable mirror images. A molecule with 1 chiral carbon atom exists as 2 stereoisomers termed enantiomers (see the example below). Enantiomers differ in their configuration (R or S) at the stereogenic center.
Are diastereomers always chiral?
Enantiomers are always chiral, but diastereomers may or may not be chiral.
Are diastereomers geometric isomers?
Geometric Isomers Exemplified Notice that geometric isomers have different physical properties. In fact, geometric isomers are diastereomers, i.e. they are stereoisomers that are not enantiomers.
What are diastereomers Slideshare?
Diastereomer Diastereomers are the stereoisomers that are not the mirror images of each other. Example: d-glucose & d-galactose are diastereomer. 17. Geometric isomerism Alkene shows geometric isomerism because it contains C=C bond which is unable to rotate. 18.What is the difference between diastereomers and enantiomers?
Enantiomers contain chiral centers that are non-superimposable & mirror images. They only come in pairs! Diastereomers contain chiral centers are non-superimposable but are NOT mirror images. There can be many more than 2 depending on the number of stereocenters.
Are diastereomers optically active?Diastereomers word is used for the compound which are optically active but are not mirror images of each other . The compounds will be same but will rotate the plane polarised light in opposite direction . Diastereomers word is used for the compound which are optically active but are not mirror images of each other .
Article first time published onAre diastereomers meso compounds?
Meso compounds are achiral (optically inactive) diastereomers of chiral stereoisomers.
Are diastereomers epimers?
Diastereomers are compounds that contain two or more chiral centres and are not mirror images of each other. … D-ribose and D-lyxose diastereomers, but they are not epimers, because they differ in configuration at both C-2 and C-3 .
Which one of the following are diastereomers?
Cis and trans are diastereoisomer of each other.
Are diastereomers constitutional isomers?
A molecule can be a constitutional isomer, diastereomer, enantiomer, and more (or none!), all at the same time to different molecules, depending on which other molecule(s) you are comparing it to.
Why do diastereomers have different properties?
The diastereomers should have DIFFERENT chemical and physical properties, including solubility, and this allows for the opportunity to separate the isomers, PRIOR to a final chiral resolution in which optical isomers are separated from each other.
How do you name diastereomers?
- The enantiomer would be (R,R)-2-methylamino-1-phenylpropan-1-ol.
- The enantiomer would be (S,R)-2-methylamino-1-phenylpropan-1-ol.
- Pseudoephedrine (S,S) and ephedrine (R,S) are diastereomers of each other.
Are stereoisomers constitutional isomers?
Constitutional (structural) isomers are compounds with the same molecular formula but with a different structure. Stereoisomers (spatial isomers) are compounds with the same molecular formula and functional structure but with a different spatial orientation of the molecules or their parts.
What are configurational stereoisomers?
Stereoisomers: Two molecules with the same constitution but different stereochemistry. … Configurational Isomers: Two molecules with the same constitution but different configuration (i.e. – same IUPAC name for constitution, different IUPAC prefix for configuration).
What are stereoisomers chemistry?
Generally defined, stereoisomers are isomers that have the same composition (that is, the same parts) but that differ in the orientation of those parts in space. There are two kinds of stereoisomers: enantiomers and diastereomers.
Do diastereomers have stereocenters?
Stereoisomers need not have stereocenters: E and Z alkenes are the common example of diastereomers. There are also classes of molecules that are chiral but have no stereocenters (we’ll be ignoring these).
What are the characteristics of diastereomers?
S.NoPropertyDiastereomers1Melting pointDifferent Melting point2Boiling pointDifferent Boiling point3SolubilityDifferent4Optical rotationDifferent values may have the same or opposite sign
Why are diastereomers chiral?
Diastereomers are stereoisomers with two or more chiral centers that are not enantiomers. Diastereomers have different physical properties (melting points, boiling points, and densities). Depending on the reaction mechanism, diastereomers can produce different stereochemical products.
What is the meaning of diastereomers?
Definition of diastereomer : a stereoisomer of a compound having two or more chiral centers that is not a mirror image of another stereoisomer of the same compound — compare enantiomer.
Which can exist both as diastereomers and enantiomers?
A [Pt(en)3]4+ FOR ANDROID YOUR ANSWER B [Pt(en)2 C1 Br]2+ C (Ru(NH3)4C12]° D [PtCl, Br2]° SOLUTION It exists as lis trans which is diastereomer and its cis form is optically active and exits as enantiomer also where as only exists.
How do you identify stereochemical relationships?
Enantiomers and diastereomers are the only two stereochemical relationships that you can have between any two molecules. The stereoisomers are any two molecules that fulfill the following two requirements: Both molecules must have the same molecular formula, and. Both molecules must have the same atom connectivity.
What is Stereoisomerism Slideshare?
Stereoisomers differ only in the way the atoms are oriented in space. They have identical IUPAC names (except for a prefix like cis or trans). They always have the same functional group(s). • A particular three-dimensional arrangement is called a configuration. • Stereoisomers differ in configuration.
Are stereoisomers mirror images of each other?
Stereoisomers are isomers that differ in spatial arrangement of atoms, rather than order of atomic connectivity. One of their most interesting type of isomer is the mirror-image stereoisomers, a non-superimposable set of two molecules that are mirror image of one another.
What is Stereoisomerism Class 11?
Stereoisomerism refers to “the isomerism that is caused by the non-similar arrangements of atoms or functional groups belonging to an atom in space”. These types of isomers have the same constitutions, but different geometric arrangements of atoms.
Is diastereomers are optically inactive?
Both of the enantiomers are diastereomers. In each case, the meso compound is not optically active, while its diastereomeric partner is optically active. It is even possible to have diastereomeric pairs in which neither member is optically active. … They are both meso compounds, and they are both optically inactive.
Are diastereomers always optically inactive?
When the enantiomeric pairs are equal amounts then the substances are said to be as optically inactive but on other hand the enantiomers contain not equal amounts then they are optically active.
Are all diastereomers optical isomers?
DiastereomersOptical isomersThey are not enantiomersOptical isomers are enantiomersSuperimposable mirror imageNon-superimposable mirror image