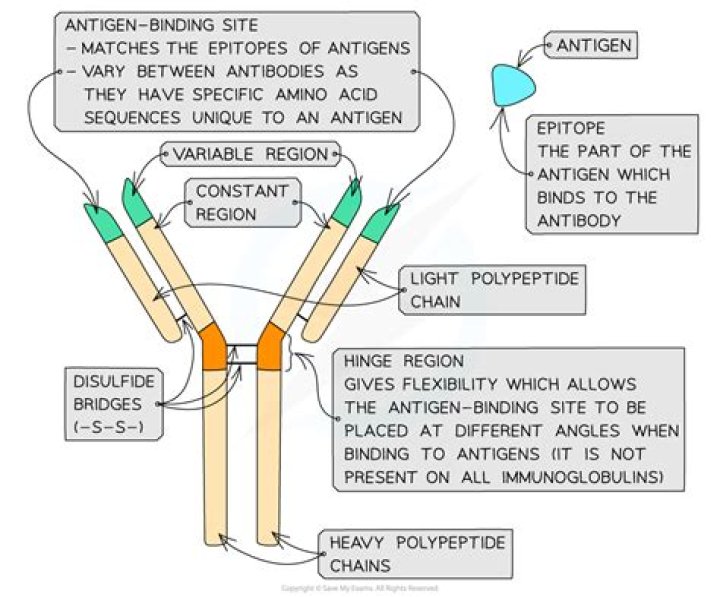
The surface of the antibody molecule formed by the juxtaposition of the CDRs of the heavy and light chains creates the site to which an antigen binds.
What are antigen binding sites made of?
The antigen-binding site of conventional immunoglobulins (Igs) is primarily composed of six complementarity-determining regions (CDRs) located in the VH and VL domains (Fig. 1A). Antibody fragments such as Fab and Fv are viewed as an autonomous unit containing a single, complete site for antigen recognition (1).
Which two structures make up the antigen binding site on an antibody?
Heavy and light chains are held together by a combination of non-covalent interactions and covalent interchain disulfide bonds, forming a bilaterally symmetric structure. The V regions of H and L chains comprise the antigen-binding sites of the immunoglobulin (Ig) molecules.
What binds to antigen binding?
The paratope is the part of an antibody which recognizes an antigen, the antigen-binding site of an antibody. It is a small region (15–22 amino acids) of the antibody’s Fv region and contains parts of the antibody’s heavy and light chains. The part of the antigen to which the paratope binds is called an epitope.What is an antibody binding site?
(A) The hinge region of an antibody molecule opens and closes to allow better binding between the antibody and antigenic determinants on the surface of an antigen. (B) Hinge flexibility also facilitates the cross-linking of antigens into large antigen-antibody complexes.
Where is the antigen binding site of this antibody quizlet?
The antigen binding site is made up of the combined variable regions of the light and heavy chains. This can be called the Fab region, and there are 2/antibody. What determines the Fc region of the antibody? The Fc region is determined by the constant region of the antibody’s genome.
How many antigen binding sites are present in an IgG?
If the affinity of the antigen-binding sites in an IgG and an IgM molecule is the same, the IgM molecule (with 10 binding sites) will have a much greater avidity for a multivalent antigen than an IgG molecule (which has two binding sites).
How do antibodies bind antigens?
Antibodies bind antigens through weak chemical interactions, and bonding is essentially non-covalent. Electrostatic interactions, hydrogen bonds, van der Waals forces, and hydrophobic interactions are all known to be involved depending on the interaction sites.What is the antigen binding?
Antigen-binding molecules. Antigen-binding molecules consist of three examples, which result in the specificity of the acquired immune response. They are able to bind to foreign antigens, and include the immunoglobulin, T-cell receptor, and major histocompatibility complex molecules.
How many antigen binding sites for antigens does each IgG antibody possess on its V regions?IgG is the most common class of immunoglobulin. It is present in the largest amounts in blood and tissue fluids. Each IgG molecule consists of the basic four-chain immunoglobulin structure—two identical H chains and two identical L chains (either kappa or lambda)—and thus carries two identical antigen-binding sites.
Article first time published onWhy are there two antigen binding sites?
The possession of two antigen-binding sites allows antibody molecules to cross-link antigens and to bind them much more stably. The trunk of the Y, or Fc fragment, is composed of the carboxy-terminal domains of the heavy chains. Joining the arms of the Y to the trunk are the flexible hinge regions.
How does antibody bind to epitope?
Antibodies bind reversibly to unique regions or epitopes within specific antigens through weak non-covalent interactions which include hydrogen, ionic, hydrophobic, and Van der Waals bonds.
Which type of cell actually secretes antibodies?
The mature B cells, called plasma cells, secrete millions of antibodies into the bloodstream and lymphatic system.
Which part's of B cell receptors form the antigen binding sites?
The V domains form the Ig antigen-binding site, whereas the C domains define the Ig isotype and its associated functions. Membrane-bound Ig (mIg) is part of the B cell receptor (BCR), soluble Ig (sIg) is secreted by plasma cells as antibody, and secretory antibodies (SIg) are polymeric sIg forms found in secretions.
Where is IgG found?
Immunoglobulin G (IgG), the most abundant type of antibody, is found in all body fluids and protects against bacterial and viral infections. Immunoglobulin M (IgM), which is found mainly in the blood and lymph fluid, is the first antibody to be made by the body to fight a new infection.
How many antigen binding sites does an immunoglobulin have quizlet?
There are two light chains and two heavy chains. The variable (Fab) region forms the antigen binding site. There is a variable region on both the heavy and light chain. There are two antigen binding sites on each antibody.
What region of the antibody determines the particular class to which an antibody belongs?
The epitope is the part of the antigen that is recognized by the antibody. In addition, the constant regions of the heavy chains determine the immunoglobulin class to which the antibody belongs ( IgM , IgG , IgD , IgA or IgE ). The immunoglobulin class is also called isotype.
What does the constant region of an antibody do?
The constant region determines the mechanism used to destroy antigen. Antibodies are divided into five major classes, IgM, IgG, Iga, IgD, and IgE, based on their constant region structure and immune function.
Which part of an antibody determines to which epitope it will bind quizlet?
In which part of the variable region of the antibody molecule does the epitope of the antigen bind? Epitope is the small region on an antigen that binds to the variable region of an antibody molecule.
What do antibodies attach to on the pathogen?
Antibodies attach to an antigen and attract cells that will engulf and destroy the pathogen. The main cells of the immune system are lymphocytes known as B cells and T cells.
What are the four types of bonds in an antibody antigen complex?
All antigen-antibody (AG-AB) bonds are weak physical bonds; covalent bonds are not encountered. The main bonds involved are: (I) Coulombic bonds; (II) Ca2+-bridges; (III) hydrogen bonds; (IV) Lifshitz–van der Waals bonds.
How many antigen binding sites for antigens does each IgG antibody possess on its V regions quizlet?
These domains are referred to as the variable region, and each antibody has two identical antigen-binding sites. The remaining domains of both H and L chains are the same in all antibodies of a given class (isotype). These domains are referred to as the constant region.
How does IgG form?
IgG antibodies are generated following class switching and maturation of the antibody response, thus they participate predominantly in the secondary immune response. IgG is secreted as a monomer that is small in size allowing it to easily perfuse tissues.
How does IgG bind?
IgG binds to cell surface receptors on many types of cells to bring about an assortment of effects: the enabling of phagocytosis (monocytes, macrophages, neutrophils) or antibody-dependent cellular cytotoxicity (monocytes, macrophages and lymphocytes) or to effect feedback control on antibody synthesis (B and T …
How many antigens can an antibody bind to?
Since an antibody has at least two paratopes, it can bind more than one antigen by binding identical epitopes carried on the surfaces of these antigens. By coating the pathogen, antibodies stimulate effector functions against the pathogen in cells that recognize their Fc region.
What is epitope binding site?
An epitope refers to the specific target against which an individual antibody binds. … Binding between the antibody and the epitope occurs at the Antigen Binding Site, which is called a paratope and is located at the tip of the variable region on the antibody.
Where are epitope located?
The small site on an antigen to which a complementary antibody may specifically bind is called an epitope or antigenic determinant. This is usually one to six monosaccharides or five to eight amino acid residues on the surface of the antigen.
What is epitope spread?
Epitope spreading (ES) refers to the development of an immune response to epitopes distinct from, and noncross-reactive with, the disease-causing epitope. Diversification, or the ability of the immune system to attack multiple targets on a pathogen has obvious advantages.
Where are B cells produced?
B lymphocytes (B cells) are an essential component of the humoral immune response. Produced in the bone marrow, B cells migrate to the spleen and other secondary lymphoid tissues where they mature and differentiate into immunocompetent B cells.
Which type of cells actually secrete antibodies Mcq?
Antibodies are produced by B-lymphocytes. These antibodies are responsible for humoral immunity, which is a type of acquired immunity. Each antibody is Y-shaped and made up of four peptide chains, namely two light chains and two heavy chains.
Which type of cell produces and secretes antibodies quizlet?
Helper T cells produce and secrete antibodies.